MECHANISMS OF RNAi AND NON-CODING RNAs
Since its relatively recent discovery, RNA interference (RNAi) has emerged as a potent, specific, ubiquitous and programmable means of gene regulation. Through a number of pathways that are conserved from S. pombe to humans, small non-coding RNAs direct molecular machinery to silence gene expression. We focus on the mechanisms and machinery that govern RNA silencing.
During RNAi, long double-stranded RNA is processed by Dicer to yield short (~19-24 nt) double-stranded RNAs that trigger the RNAi response. These short RNAs get incorporated into effector complexes called the RNA Induced Silencing Complex (RISC). The short RNA (siRNAs or miRNAs) then acts to guide the RISC complex to its target through base complementarity. In all cases, these complexes contain a small single-stranded RNA and an Argonaute protein.
ARGONATUE AND FRIENDS
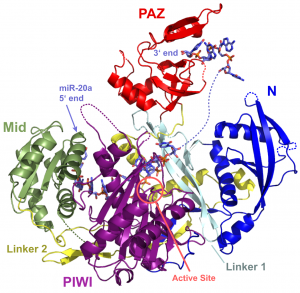
Several years ago, we determined the first structure of a full-length Argonaute protein (link to PDB or structural model and paper). This archael protein was composed of four domains: N, PAZ, Mid and PIWI. The structural similarity of the PIWI domain to RNase H-like nucleases revealed Argonaute as the key effector protein in the RISC complex as it was identified as Slicer – the enzyme that produces the endonucleolytic cut in an mRNA when the guide and target mRNA are fully complementary. This places Argonaute at the heart of this pathway.
With our ability to reproducibly purify unbound human Argonautes 1 and 2 (hAgo1 & hAgo2), we were able to specifically load these Argonautes with discrete miRNAs, and determined structures of these complexes (links). We showed how miRNAs bind extensively to Argonautes and detailed the effect they have on the protein’s stability. We were also able to convert a catalytically inactive, non-slicing Argonaute (hAgo1) to an active slicer.
We continue to investigate how Argonautes function as effectors that influence gene expression in processes as diverse as transcriptional gene silencing, post-transcriptional gene silencing, translation and even regeneration.
Relevant Pubs
B. Bibel, E. Elkayam, S. Silletti, E.A. Komives and L. Joshua-Tor, Target binding triggers hierarchical phosphorylation of human Argonaute-2 to promote target release, bioRxiv, doi: https://doi.org/10.1101/2022.01.06.475261 (2022). Preprint.
J. Li, C. Wu, W. Wang, Y. He, E. Elkayam, L. Joshua-Tor and P. T. Hammond, Structurally Modulated Co-delivery of siRNA and Argonaute 2 for Enhanced RNA Interference, Proc. Natl. Acad. Sci., USA, 115 (12), E2696-E2705 (2018).
E. Elkayam, C.R. Faehnle, M. Morales, J. Sun, H. Li and L. Joshua-Tor, Multivalent recruitment of human Argonaute by GW182, Mol. Cell, 67 (4), 646-658 (2017).
E. Elkayam, R. Parmar, C. R. Brown, J. L. Willoughby, C. S. Theile, M. Manoharan*, L. Joshua-Tor*, siRNA carrying an (E)-vinylphosphonate moiety at the 5’ end of the guide strand augments gene silencing by enhanced binding to human Argonaute-2, Nucl. Acids Res. (2016).
C. R. Faehnle*, E. Elkayam*, A. D. Haase, G. J. Hannon and L. Joshua-Tor, The making of a Slicer: activation of human Argonaute-1, Cell Rep, 3, 1901-1909 (2013).
E. Elkayam, C.-D. Kuhn, A. Tocilj, A. D. Haase, E. M. Greene, G. J. Hannon and L. Joshua-Tor, The structure of human Argonaute-2 in complex with miR-20a, Cell, 150, 100-110 (2012).
E. Yigit, P. J. Batista, Y. Bei, K. M. Pang, C. G. Chen, N. H. Tolia, L. Joshua-Tor, S. Mitani, M. J. Simard and C. C. Mello, Analysis of the C. elegans Argonaute family reveals that distinct Argonautes act sequentially during RNAi, Cell, 127(4), 747-57 (2006).
D. V. Irvine*, M. Zaratiegui*, N. H. Tolia*, D. B. Goto, D. H. Chitwood, M. W. Vaughn, L. Joshua-Tor and Robert A. Martienssen, Argonaute slicing is required for heterochromatic silencing and spreading, Science, 313, 1134-1137 (2006).
F. V. Rivas*, N. H. Tolia*, J. J. Song*, J. P. Aragon, J. Liu, G. J. Hannon** and L. Joshua-Tor**, Purified Argonaute2 and an siRNA form recombinant Human RISC, Nature Struct. Mol. Biol., 12 (4), 340-349 (2005)
J. J. Song, S. K. Smith, G. J. Hannon and L. Joshua-Tor, Crystal structure of Argonaute and its implications for RISC Slicer activity, Science, 305, 1434-1437 (2004)
J. Liu, M. A. Carmell, F. V. Rivas, C. G. Marsden, J. M. Thomson, J. J. Song, S. M. Hammond, L. Joshua-Tor and G. J. Hannon, Argonaute2 is the catalytic engine of mammalian RNAi, Science, 305, 1437-1441 (2004)
J. J. Song, J. Liu, N. H. Tolia, J. Schneiderman, S. K. Smith, R. A. Martienssen, G. J. Hannon and L. Joshua-Tor, The crystal structure of the Argonaute2 PAZ domain reveals an RNA binding motif in RNAi effector complexes, Nat. Struct. Biol., 10 (12), 1026-1032 (2003).
Human Argonaute-2 in complex with miR20a
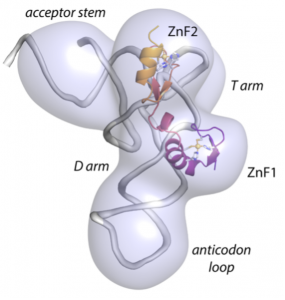
Cryo-EM structure of mouse Arx/Gtsf1 in complex with tRNA. Gtsf1 interacts directly with tRNA and is positioned such that ZnF1 interacts with the tRNA D arm. ZnF2 extends toward the tRNA acceptor stem.
Relevant Pubs
J.J. Ipsaro, P.A. O’Brien, S. Bhattacharya, A.G. Palmer III and L. Joshua-Tor, Asterix/Gtsf1 links tRNAs and piRNA silencing of retrotransposons, Cell Reports 34 (13), 108914 (2021).:
C. Stein*, P. Genzor*, S. Mitra*, A. R. Elchert*. L. Benner, S. Sobti, J. J. Ipsaro, M. Hammell, L. Joshua-Tor and A. D. Haase, Decoding the 5’ nucleotide bias of PIWI-interacting RNAs (piRNAs), Nature Comm., doi: 10.1038/s41467-019-08803-z (2019).
J. J. Ipsaro*, A. D. Haase*, S. R. Knott, L. Joshua-Tor** and G. J. Hannon**, The structural biochemistry of Zucchini implicates it as a nuclease in piRNA biogenesis, Nature, 491, 279-283 (2012).
RNAi IN THE GERMLINE
Another clade of Argonautes, the Piwi proteins are of particular interest since they associate with a specific class of small RNAs, the piRNAs. These molecules reside in germ cells and protect their genomes against instability caused by mobile genetic elements. Unlike siRNAs and miRNAs whose biogenesis depends on Dicer, primary piRNA biogenesis is independent of Dicer. Rather, long ssRNA transcripts are parsed into smaller 5’- monophosphorylated fragments. The piRNA precursor then appears to be loaded into its Piwi-family partner. The 3’ ends of the piRNAs are then formed by a Mg2+-dependent trimming activity. piRNAs can be amplified in a feed-forward ‘ping-pong’ mechanism, carried out by two different Piwi proteins, generating so-called secondary piRNAs. Once mature piRNAs are loaded into Piwis, the resulting effector complexes are able to direct silencing at either the transcriptional or post-transcriptional level.
We showed that an enzyme called Zucchini is a single strand-specific nuclease (link). Our crystal structure of mouse Zucchini (mZuc) revealed a long, positively-charged groove with zinc finger motifs at each end that can accommodate single-stranded nucleic acid substrates. mZuc displays a striking architectural similarity to HKD nucleases and is consequently dissimilar to HKD lipases. These findings implicate Zucchini as a primary piRNA biogenic nuclease perhaps being responsible for generating the 5’ ends of primary piRNAs.
By using structural biology, biochemistry, biophysical techniques, along with functional studies, we seek to determine the role of various factors that are critical for gene silencing in the germline.
For more, please watch Jon Ipsaro’s talk on Asterix/GTSF1 here.
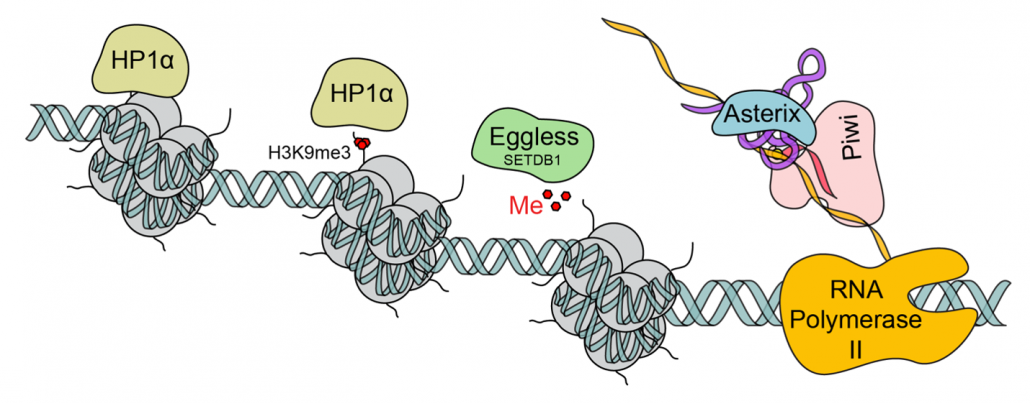
Model for the role of Asterix/Gtsf1 in silencing. In the nucleus, Asterix may utilize tRNAs to recognize primer binding sites in nascent transposon transcripts. Enhanced recruitment of piRISCs (pink) could then be achieved through interactions with Asterix’s C-terminal tail in addition to the protein-protein interactions between Asterix and Piwi, and the piRNA with the transcript. This leads to further recruitment of histone modification enzymes (such as Eggless/SETDB1; green) and eventual binding of HP1α (beige) to establish heterochromatin.
THE let-7 PATHWAY
Let-7 is among the first miRNAs discovered, where it is involved in controlling cell fate during late stage larval development in C. elegans. The let-7 family is broadly expressed in somatic cells and regulates cellular proliferation and differentiation, as well as the repression of several oncogenes and key regulators of mitogenic pathways. The Lin28 protein was discovered to be the post-transcriptional regulator of let-7 biogenesis. Lin28 is highly expressed in embryonic stem cells and is naturally active in early development where it suppresses the expression of mature let-7 to maintain pluripotentcy. The Lin28-let-7 pathway has been described as a bistable switch. Each molecule represses expression of the other, and once the cell changes its state, the result is differentiation, or if the switch is reversed, cancer.
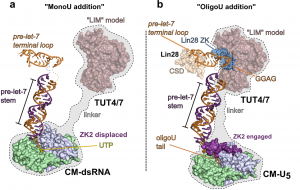
In differentiated cells, TUT4 and TUT7 promote let-7 processing by monouridylating certain let-7 precursors (pre-let-7) that carry a single 3’-nucleotide overhang, rather than the more Dicer-ready two-nucleotide overhang pre-miRNAs. However, in stem cells, Lin28 binds to pre-let-7, which triggers 3’ oligouridylation by TUT4/7. The oligoU tail serves as a decay signal that is rapidly degraded by Dis3L2. We determined the structure of Dis3L2 in complex with an oligoU RNA to mimic the uridylated tail of pre-let-7. The RNA-binding domains form an open funnel on one face of the catalytic domain that allows RNA to navigate a path to the active site. An extensive network of uracil-specific interactions span the first 12 nucleotides of the oligoU-tailed RNA, explaining how Dis3L2 recognizes, binds and processes uridylated pre-let-7 in the final step of the Lin28-let-7 pathway. We continue to investigate this important and fascinating pathway.
Relevant Pubs
K. Meze, A. Axhemi, D.R. Thomas, A.Doymaz and L. Joshua-Tor, A shape-shifting nuclease unravels structured RNA, bioRxiv, https://doi.org/10.1101/2021.11.30.470623 (2021).
C. R. Faehnle*, J. Walleshauser*, and L. Joshua-Tor, Multi-domain utilization by TUT4 and TUT7 in control of let-7 biogenesis, Nat. Struct. Mol. Biol., 24 (8), 658-665 (2017).
C.R. Faehnle*, J. Walleshauser*, and L. Joshua-Tor, Mechanism of Dis3L2 substrate recognition in the Lin28/let-pathway, Nature, 514, 252-256 (2014).
Our model of the TUT4(7) activity switch. (a) MonoU addition (biogenesis mode): Group II miRNAs are monouridyated by the CM through recognition of the 1 nt overhang end structure. The LIM is a homology model built using the CM structure as a template (light pink, transparent surface). The pre-let-7 terminal loop is rendered from the structure of the Lin28/pre-let-7g complex (orange cartoon) (PDB ID 3TS2). (b) Lin28-dependent oligoU addition (destruction mode). Based on data from our lab and work reported by the Piotr Sliz lab, the Lin28 CCHCx2 domain (blue surface) recruits the LIM of TUT4(7) to pre-let-7 to form a stable ternary complex, that turns on progressice oligoU addition by the CM, aided by direct engagement of ZK2.
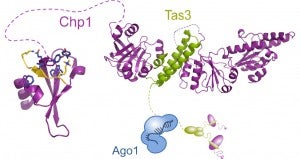
Chp1 and Tas3 form a bridge between chromatin and the RNAi machinery. The chromodomain (CD) of Chp1 binds H3K9 methyl marks on nucleosomes, while the GW domain of Tas3 binds to Ago1.
RNAi AND HETEROCHROMATIN
RNAi pathways are also used to repress genes at the transcriptional level by guiding heterochromatin formation. These pathways are most fully characterized in in the yeast S. pombe where heterochromatin is essential for kinetochore assembly and consequently for proper chromosomal segregation during cell division. Assembly of centromeric heterochromatin requires the RITS complex, a specialized RISC, which physically anchors small RNAs to chromatin. It consists of the Argonaute protein Ago1, the chromodomain protein Chp1, a GW-like protein Tas3 and siRNAs derived from centromeric repeats. We characterized Chp1’s high-affinity binding to chromatin and showed it to be critical for establishment of centromeric heterochromatin. Our structure of the C-terminal half of Chp1 with the Tas3 N-terminal domain, revealed the presence of a PIN domain in Chp1, and showed that it contributes to post-transcriptional gene silencing of subtelomeric transcripts independent of RNAi. We suggested that Chp1-Tas3 provides a solid and versatile platform to recruit both RNAi-dependent and RNAi-independent gene-silencing pathways for locus-specific regulation of heterochromatin, and that the reach of long and flexible regions in these two proteins might be important for contacting other nucleosomes at these loci. The RITS complex recruits the H3K9 methylase, Clr4, which is part of a large complex called CLRC. We are elucidating the functions of components of this complex.
Relevant Pubs
C. Kuscu*, M. Zaratiegui, H. S. Kim, D. A. Wah, R. A. Martienssen, T. Schalch* and L. Joshua-Tor*, The CRL4-like Clr4 Complex in S. pombe depends on an exposed surface of Dos1 for heterochromatin silencing, Proc. Natl. Acad. Sci., USA, 111 (5), 1795-1800 (2014).
T. Schalch, G. Job, S. Shanker, J. F. Partridge and L. Joshua-Tor, The Chp1-Tas3 core is a multifunctional platform critical for gene silencing by RITS, Nat. Struct. Mol. Biol., 18, 1351-1357 (2011).
T. Schalch, G. Job, V. J. Noffsinger,S. Shanker,C. Kuscu,L. Joshua-Tor*,and J. F. Partridge*, High-affinity binding of Chp1 chromodomain to K9 methylated histone H3 is required to establish centromeric heterochromatin, Mol. Cell, 34, 36-46 (2009).